Find a Study
Opportunities for patients and families to get involved with research including:
- Becoming an patient advisor to a research study
- Participating in focus groups
- Taking a survey
- Participating in a disease registry
- Enrolling as a clinical trial participant
Current opportunities to get involved with research
Does your toddler have a peanut allergy?
If your child has or may have a peanut allergy, we ask you to consider the COMFORT Toddlers study. It may help us learn more about treating peanut allergy in toddlers.
Struggling to manage chronic urticaria symptoms (hives)?
Consider enrolling in the BLU-808-1201 clinical research study. You may qualify if you have chronic spontaneous urticaria and are still experiencing symptoms such as hives even after taking antihistamines. Participation lasts at least 4 months and includes the following:
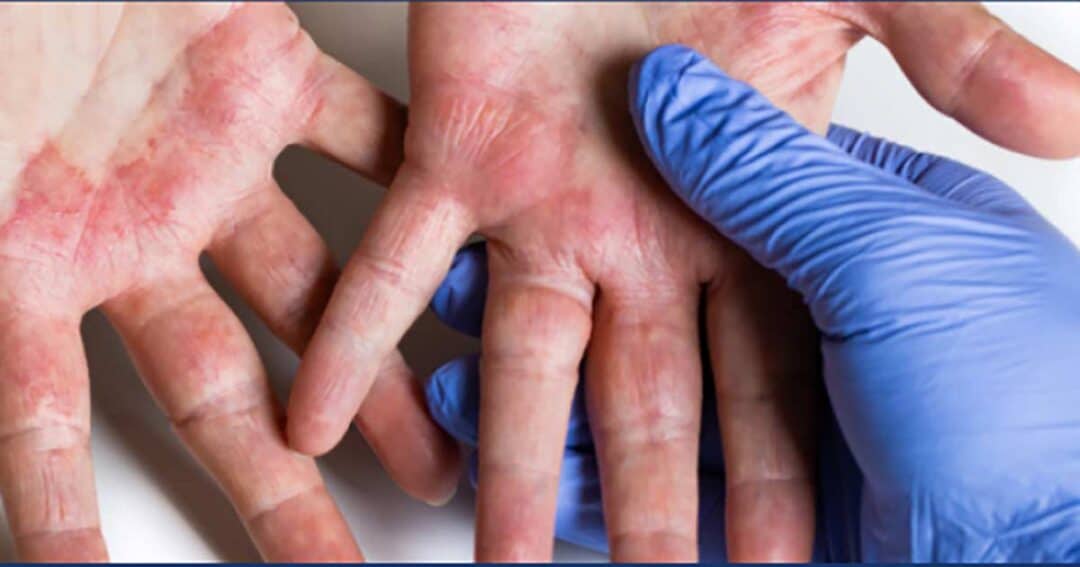
Be Part of Progress in Eczema Research
Who may be a good candidate for this atopic dermatitis (eczema) study? You may consider applying to be a participant if you meet the following criteria for qualification
Asthma Research Study
About the RAINER Asthma Research Study This study is testing an investigational drug called solrikitug to see if it is safe and helps adults with asthma. The medication works by stopping the body’s immune system from causing swelling and asthma flare-ups. Asthma can cause trouble breathing, coughing, wheezing, chest tightness, tiredness, and feeling worried. The RAINIER study…
Chronic Hives Study
The focus of this study is to evaluate the efficacy of an investigational drug, EVO756, in adults with moderate to severe CSU, and how safely it interacts with the body. Participants will receive either the study drug or placebo in tablet form to take for 12 weeks and be evaluated on an outpatient basis. The…
Chronic Spontaneous Urticaria (CSU) At-Home Research Opportunity
Sanguine Biosciences, a provider of at-home clinical research services, will share an at-home study opportunity with those interested in advancing chronic spontaneous urticaria (CSU) research.
Opportunities for patients and families to get involved with research including:
- Becoming an patient advisor to a research study
- Participating in focus groups
- Taking a survey
- Participating in a disease registry
- Enrolling as a clinical trial participant